Biomarkers of Spinal and Bulbar Muscle Atrophy (SBMA): A Comprehensive Review
Biomarkers in SBMA
A biomarker is a parameter that can be measured accurately and reproducibly and used as an indicator of normal biological processes, pathogenic processes, or pharmacologic responses to a therapeutic intervention (WHO definition, 1998). An ideal biomarker should have a predictive value and capture subtle changes over relatively short periods of time. Additional requirements to biomarkers include cost-effectiveness, non-invasiveness, and reproducibility. It is generally agreed that no single biomarker is suitable for diagnostic, prognostic and monitoring roles and a panel of several markers may be better suited as multirole indicators. SBMA is a rare and slowly progressing condition, therefore the development of sensitive outcome measures would enable smaller sample-size and shorter duration of pharmaceutical trials.
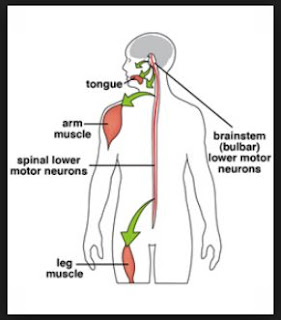
Biomarkers of Neurological Involvement in SBMA
In recent years, an unprecedented interest has developed in the standardized assessment of neuromuscular performance in SBMA, evaluation of novel therapeutic strategies and in the launch of national SBMA registries. Many of the commonly used instruments, such as the MRC score, respiratory function parameters, the modified Norris scale, ALSFRS-r, Quantitative Myasthenia Gravis Score etc. are non-specific to SBMA, yet remain widely utilized. As these tools have been developed for other conditions, new batteries of tests have been recently proposed to specifically appraise disability in SBMA.
Discussion and Future Perspectives
Interest in SBMA biomarkers has grown steadily in recent years, fuelled both by accruing knowledge about pathogenesis and novel therapeutic strategies. SBMA is now widely recognized as a multisystem syndrome. A multitude of studies focus on multi-organ involvement, and the systemic phenotype is now considered just as relevant as the neurological manifestations. It is increasingly recognized that non-neurological features of the disease have an equally important impact on the patients' quality of life. Until now, clinical trials on SBMA focused almost exclusively on the treatment of motor symptoms, but a shift to targeted molecular therapies and focus on systemic processes are likely to be witnessed in the near future. From a clinical trial perspective, ideal biomarkers should undergo robust validation, sensitivity and specificity profiling, and sampling and measurement harmonization across different centers. Crucially, candidate markers should be able to detect the subtle changes expected after the administration of a specific treatment. Given the particularly slow progression rates observed in SBMA, the definition of an effective outcome measures is challenging. The integration of neurological, metabolic, and endocrine indicators seems essential into composite biomarker panels in addition to functional scales. Serum creatinine levels appear to correlate strongly with motor impairment and HOMA-IR index with disease duration. The convincing validation of these parameters and their use as effective outcome measures in clinical trials will require robust multicenter study designs.

No comments:
Post a Comment
Please feel free to comment. By taking a moment to share your thoughts you add much to these articles. The articles then become more than just something I said or believe. In addition, by adding a comment, you might just be helping the next reader by sharing your opinion, experience, or a helpful tip. You can comment below or by sending me an email. I look forward to hearing from you.